In this blog we share learning from a screening safety incident. The shared learning is specifically aimed at laboratories that provide Down’s, Edwards’ and Patau’s screening. Screening laboratories use specialist software to combine biochemical results with other information, such as ultrasound measurements and maternal age, to come up with the level of chance that the unborn baby has one of these conditions.
It is important that the calculations are accurate, especially when they are affected by many other factors. Internal quality control (IQC) processes are used to detect and correct problems so that pregnant women can be confident in the results they are given. We want to highlight here the value of monitoring MoM (multiple of medians) values in laboratories as part of these processes.
We have a national quality assurance process for Down’s, Edwards’ and Patau’s screening called the Down’s syndrome Screening Quality Assurance Support Service (DQASS) that provides 6-monthly retrospective feedback to screening laboratories. Laboratories also need their own IQC processes.
Screening for Down’s, Edwards’ and Patau’s syndromes relies on standardising biochemical concentrations into MoM (multiples of the median) values. A MoM value of 2, for example, means that the concentration is twice as high as the median value among pregnancies with specific characteristics.
Standardisation is done for factors including gestational age, maternal weight, ethnicity and smoking, as illustrated in Figure 1. Marker concentrations are standardised into MoM values using calculations embedded within the screening software.
Biases in these MoM values will impact on the screening results and the overall performance of the test. DQASS only provides diagnostic feedback on MoM values retrospectively, so there needs to be a real time process of monitoring MoM values within laboratories.
Here, we highlight the benefits of continuous monitoring of MoM values using cumulative sum (CUSUM) charts of log transformed MoM values. Large biases can be identified with a relatively small sample of data, such as a single day’s run.
Detection of more moderate biases needs more data. The diagnostics spreadsheet used to create the CUSUM figures below has been shared with all laboratories that provide Down’s, Edwards’ and Patau’s screening.
Case study: timely identification of large biases
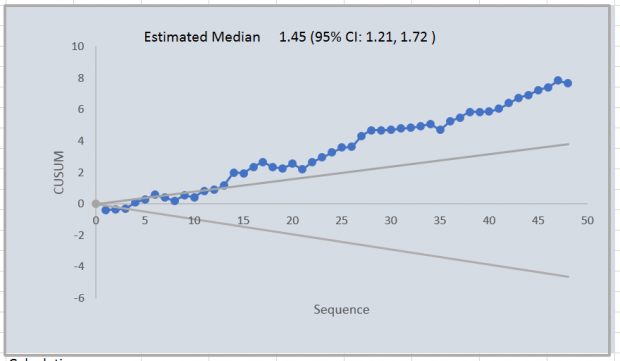
Suppose that a problem resulted in a substantial positive bias of around 40% in the hormone human chorionic gonadotrophin (hCG β) for a run of 48 samples, and that this was not identified.
A CUSUM plot of the log MoM for this run is shown in Figure 2. The positive bias is shown by the increasing trend of the CUSUM. The slope can be compared with the reference lines shown in grey for median MoM values of 0.9 and 1.1 and shows a consistent bias far in excess of 10%. The estimated median MoM for the run is 1.45 with a confidence interval ranging from 1.21 to 1.72.
The information provided by the CUSUM and the confidence interval would alert the laboratory to a potentially serious problem and the need for further investigation before the results from this run were released to women.
Case study: longer term monitoring for moderate biases
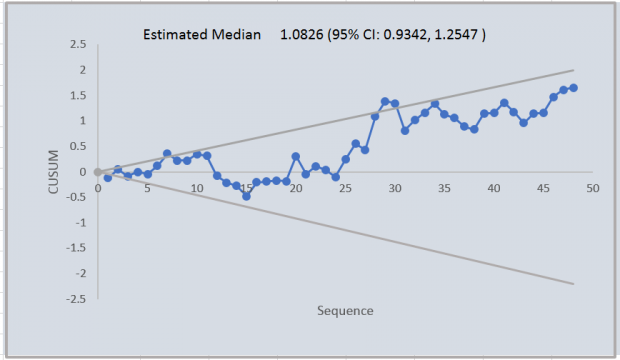
Figure 3 shows a CUSUM chart for a run of 48 samples with a bias of around 10%. Although there is an increasing trend there is not enough evidence to justify a change as the deviation from target could be explained by chance.
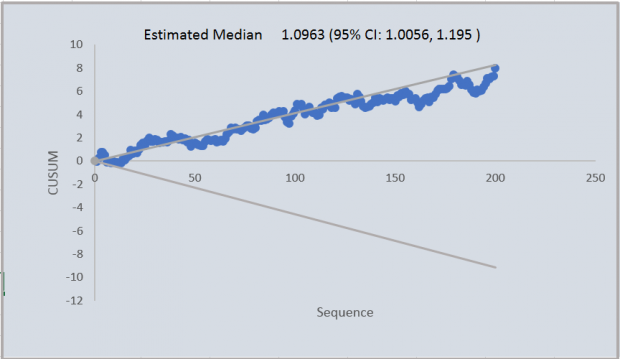
If the process is continued and 200 observations are shown, the bias becomes apparent – as shown in Figure 4.
These examples demonstrate that monitoring MoM values in real time for small runs is an effective way of detecting large biases in a timely way and that longer term monitoring of MoM values is an effective way of identifying more moderate biases.
We are therefore continuing to encourage screening laboratories to monitor MoM values in laboratories as part of their routine IQC processes.
PHE Screening blogs
PHE Screening BLOGs provide up to date news from all NHS screening programmes. You can register to receive updates direct to your inbox, so there’s no need to keep checking for new blogs.
2 comments
Comment by S Schlanker posted on
I believe that both Nadia and David are both interesting and skilled lecturers - as I’ve heard them ; I understand screening on a day to day level but this article is confusing and doesn’t tell a clinician working with patients anything of any benefit - learning from an incident is always good but it is important to know what we can control NOT what is out of our control
Comment by Mike Harris posted on
Thank you for your comment.
We appreciate that this blog is trying to convey some complicated information which is challenging. The blog and learning are specifically aimed at biochemists and other laboratory staff working in this field. We believe that they will be able to interpret and take the appropriate actions and learning forward. There are no specific actions for clinicians working with patients.
Kind regards,